How Much Do You Need to Know About Mass Spectroscopy for Mcat
Spectroscopy for the MCAT: Everything You Need to Know
/
Learn key MCAT concepts about IR and NMR spectroscopy, plus practise questions and answers

(Note: This guide is function of our MCAT Organic Chemistry series.)
Tabular array of Contents
Part ane: Introduction to IR and NMR spectroscopy
Part two: IR spectroscopy
a) Experimental procedure
b) Key functional groups
c) Case
d) Related forms of spectroscopy
Office 3: NMR spectroscopy
a) Experimental procedure
b) Reading a spectrum
c) Splitting and coupling
d) Example
Part four: High-yield terms
Function v: Passage-based questions and answers
Part 6: Standalone questions and answers
Part 1: Introduction to IR and NMR spectroscopy
Since molecules are so small, how do chemists determine the construction of an unknown molecule? If researchers cannot detect single molecules with the naked eye, how do we verify the identities of chemical products and reactants?
The answer lies in spectroscopy and other experimental techniques. In this guide, nosotros'll cover everything you need to know about ii of the most important forms of spectroscopy: infrared (IR) and nuclear magnetic resonance (NMR) spectroscopy. From the fundamental experimental principles to how to interpret the resulting data, by the stop of this guide, you lot'll be equipped for anything the MCAT has in store for you come test mean solar day.
Note that while you may have learned well-nigh IR and NMR spectroscopy in your undergraduate courses, the level of depth required to do well on these topics for the MCAT is significantly lower. For example, you may have been asked to describe a molecule that generated a certain spectrum in an undergraduate organic chemical science lab course. The MCAT will non require this gratuitous recall and product of information—afterwards all, the MCAT is a multiple-pick exam.
With that all out of the way, let's brainstorm!
Office 2: IR spectroscopy
a) Experimental procedure
Infrared spectroscopy, or IR spectroscopy, involves the casting of infrared light through a molecular sample. The energy from the light then causes covalent bonds inside the molecule to vibrate. These bonds can vibrate by stretching and angle.
Every bit the calorie-free passes through the sample, a spectrometer measures the percentage of light that passes through the other finish. This percentage is expressed as the percent transmittance of lite over a range of frequencies. The frequencies at which the calorie-free is absorbed depend on the types of functional groups present in the sample. Thus, IR spectroscopy can place specific functional groups and types of bonds in a compound.
One way to visualize the vibrations of the covalent bonds is by using the spring model. Imagine two balls connected by a spring. When the tension of the spring is increased, the frequency of its vibration volition increase. Additionally, when the forcefulness of the bond is increased, the frequency at which it vibrates volition increase. Lastly, if the mass of the two balls is increased, the frequency will subtract.
These are the aforementioned principles behind tuning a guitar: the tighter a string is tuned, the higher its pitch will be. When the forcefulness of a guitar string is increased, a thick string is used instead of a sparse cord, the frequency also changes. Thicker strings, like those you'll find on a bass guitar, produce lower frequencies than the thinner strings you'll find on a regular guitar.

Effigy: Stretching and bending of covalent bonds betwixt atoms, every bit visualized by the spring model.
These intramolecular vibrations and rotations are elicited and detected with IR spectroscopy.
b) Central functional groups
The plot of an IR spectrum has percent transmittance on the y-axis and the wavenumber on the x-axis.
Wavenumber is measured in cm-1 and functions equally an analog for the frequency of light that is used. Perhaps counterintuitively, a lower percent transmittance on the spectrum indicates the presence of a sure functional group. Additionally, the wavenumber decreases to the rightward direction on IR spectra.
The fingerprint region is a special range of wavenumbers from around 1500 to 500 cm-1 that is unique to a certain compound. For the MCAT, you'll exist working within a different range, from around 4000 to 400 cm-one. The tabular array below lists the cardinal functional groups for which y'all should memorize their characteristic wavenumbers. These, in particular, are important because they are characteristic of biological compounds.
| Functional Group | Wavenumber |
|---|---|
| | |
| | |
| | |
| | |
For more data on the mode they bear in reactions, be sure to refer to our guide on important functional groups.
c) Example
Let's take what has been discussed so far and employ it using an instance trouble. Try to identify the functional groups present in the IR spectrum below.
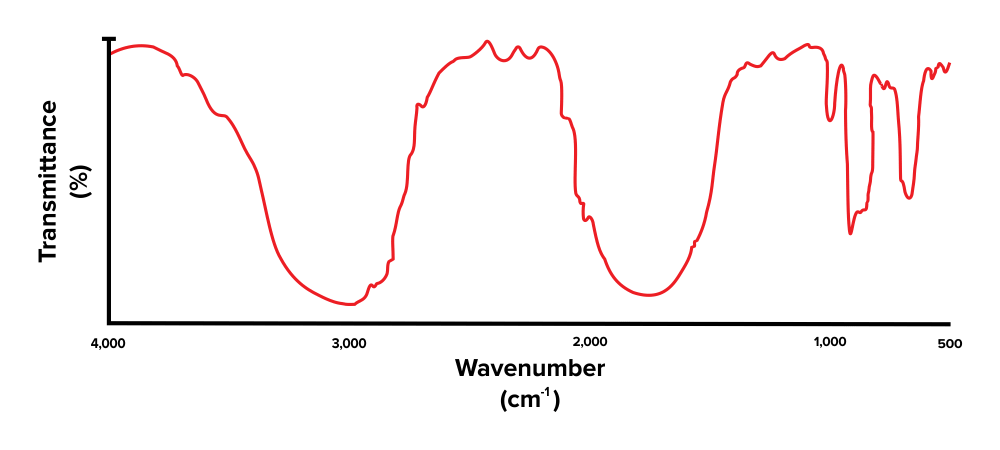
Figure: An unlabelled IR spectrum.
Recall that a lower percentage transmittance indicates the presence of the corresponding functional group. When interpreting an IR spectrum, be sure to focus on the dips and minima in the spectrum (rather than the peaks and maxima).
Commencement, note that there is a broad peak at around 3,300 to 3,000 cm-ane. This corresponds with the presence of an alcohol functional grouping (O-H). Adjacent, there is some other height, present effectually the 1800 to 1600 cm-one range. This indicates that the compound has a carbonyl grouping (C=O). The peaks we see towards the very right are within the fingerprint region and are out of scope for the MCAT.
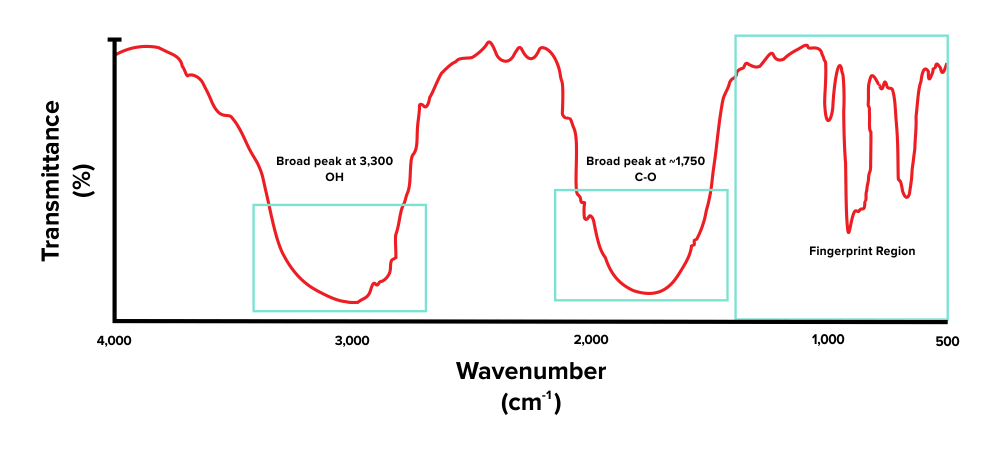
Figure: An IR spectrum of a molecule that contains -OH and C=O functional groups.
On the MCAT, yous may be asked to identify the molecular compound that has generated a corresponding IR spectrum. Permit's try an example, referring to the IR spectrum we've been working with.
Which of the following structures is most probable the chemical compound that generated the IR spectrum?
A)
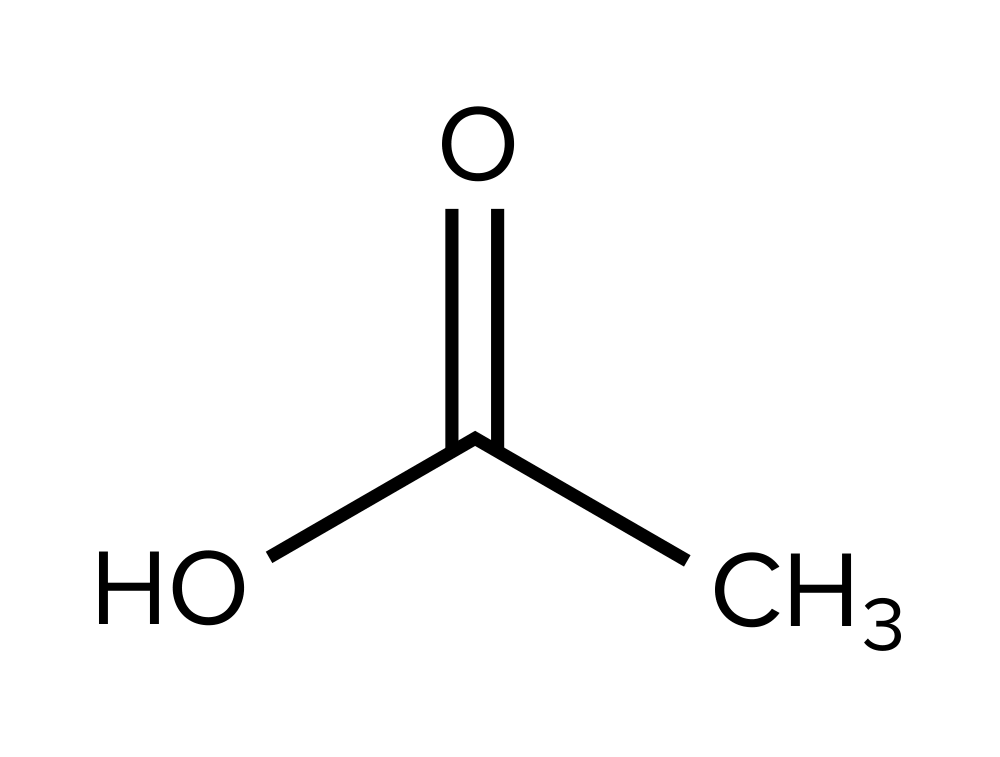



The all-time way to solve these, specially when given more complex structures, is by process of elimination. Notation that the molecule presented in selection C has a distinctive -NH2 grouping. This should generate a sharp peak at around ~1,750 cm-ane. However, since this is non present in the IR spectrum, reply pick C can be eliminated.
Further, annotation that choice D is unique because information technology is the just one lacking a carbonyl group. Since the IR spectrum has confirmed the presence of a C=O bond in the molecule (a sharp peak at effectually ~1,750 cm-ane), this cannot be the right construction.
Finally, choices A and B differ in the presence of an alcohol group. Since the IR spectrum has confirmed the presence of an -OH bond in the molecule (a broad peak at ~3,000 cm-1), choice B cannot be the correct construction. Therefore, choice A, which contains a carboxylic acid, is the only molecule presented in the answer options that could take generated the IR spectrum.
d) Related forms of spectroscopy
While spectroscopy using light from the infrared region is widely used, other frequencies of light may besides be used for spectroscopy.
Spectroscopy using visible light can be useful to detect the change in concentration of a colored indicator. Indicator molecules can be used to measure the progress of a reaction. For instance, the dye malachite green binds to free inorganic phosphate to turn a vibrant greenish. The dye will absorb light at green wavelengths while transmitting light at a red wavelength, since ruby and light-green are complementary colors.
Spectroscopy using ultraviolet light can be useful for detecting conjugated electron systems within aromatic molecules. These ultraviolet wavelengths interact with pi-electron and nonbonding electron transitions to produce dissimilar frequencies of transmitted light.
Part 3: NMR spectroscopy
a) Experimental procedure
Nuclear magnetic resonance spectroscopy, or NMR spectroscopy, is a blazon of spectroscopy that allows scientists to determine the construction of a molecule, including the presence of central functional groups. This technique takes advantage of the fact that the nucleus of an atom can be in one of many different magnetic states when placed in an external magnetic field. Ane magnetic state corresponds with alignment with the magnetic field and low energy, while the other corresponds with unalignment with the magnetic field and higher energy.
Molecular samples are placed in a magnetic field and exposed to different frequencies of low-cal, the absorption of which can crusade shifts between magnetic states. Depending on atoms present in the molecule and its magnetic environment, absorption will occur at different frequencies. Measuring this absorption at unlike frequencies thereby allows the conclusion of any elements present, along with the structure of the molecule.
b) Reading a spectrum
NMR spectroscopy plots list frequency on their y-axis and chemical shift (δ), measured in parts per 1000000, on the 10-axis. Similar to IR plots, this value decreases the further right you continue an NMR spectrum.
A standard molecule known equally tetramethylsilane (TMS) calibrates the spectra and sets a reference indicate. Thus, the δ of TMS is 0 ppm. TMS has the chemical formula Si(CHthree)four.
Moving toward the left of an NMR spectroscopy plot (toward higher chemical shifts) is referred to as going downfield. Moving toward the correct of an NMR spectroscopy plot (toward lower chemical shifts) is referred to every bit going upfield. This terminology is important when discussing deshielding.
Deshielding is a phenomenon that occurs when electron-withdrawing groups pull electrons away from the core of an cantlet, making it more susceptible to the magnetic field. Electron-withdrawing groups tend to contain elements of very high electronegativity. (For more data on this, be sure to refer to our guide on periodic trends.) This "pulling abroad" of electron density makes the nucleus more susceptible to magnetic shifts, thus moving a meridian further downfield. Conversely, shielding occurs when electron-donating groups increase the electron density virtually the core of an atom, reducing its susceptibility to the magnetic field. This moves a peak further upfield.
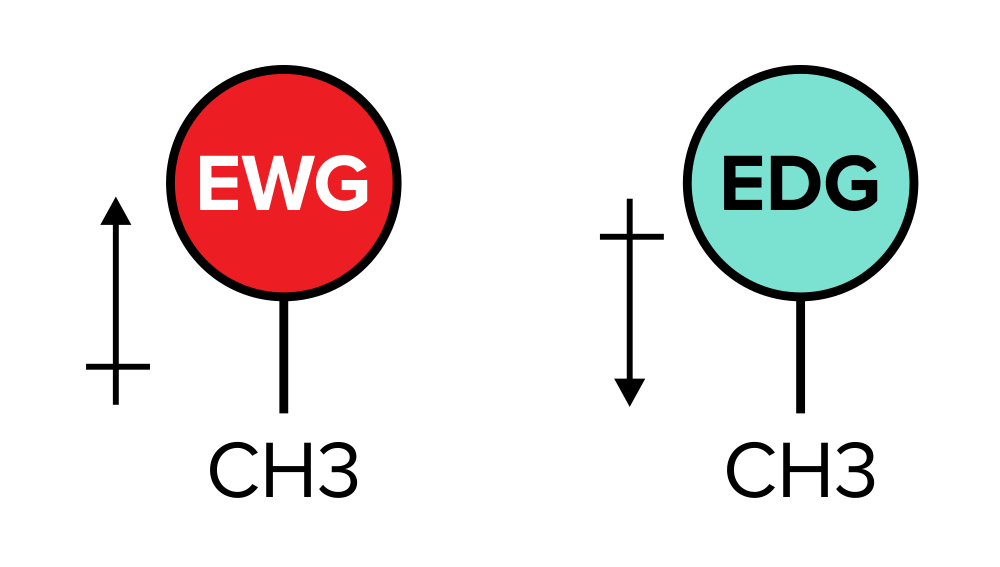
Figure: Electron-withdrawing groups vs electron-altruistic groups
On test day, y'all will most likely encounter problems involving H-NMR spectroscopy. For this type of spectroscopy, certain types of bonded protons (in the form of hydrogen atoms) cause a specific chemical shift. The tabular array below summarizes the chemical shifts you should memorize and know.
| Blazon of proton | Chemical shift / δ (ppm) |
|---|---|
| | |
| | |
| | |
| | |
| | |
| | |
| | |
c) Splitting and coupling
Spin-spin splitting, also known as coupling, occurs when hydrogen atoms fastened to adjacent carbons interfere with 1 some other's magnetic environment. These protons, which are often located in the same chemical environment and bonded to the aforementioned heteroatoms, are considered to be equivalent protons. The coupling abiding (J) is used to define the forcefulness of these magnetic interferences. It is measured in Hertz. Ultimately, the presence of splitting of protons in a magnetic field is useful because information technology results in distinguishing features between isomers.
Depending on the number of adjacent carbons, any peak on an NMR spectrum may be dissever up into subpeaks. We tin predict the number of subpeaks that will arise using the formula, n + ane, where n is equal to the number of hydrogen atoms that are iii bonds away from the hydrogen cantlet of involvement. In other words, the carbon atoms that the hydrogen atoms are bonded to are neighboring.
If 2 subpeaks result on the spectrum, the trace is referred to as a doublet. If three subpeaks result, they are referred to as a triplet. A set of whatsoever greater number of subpeaks is referred to as a multiplet.
d) Case
Permit'due south take what we've discussed and employ information technology using an example.
Given the compound below and its 1H-NMR spectrum, make up one's mind which sets of hydrogen atoms represent with each tiptop.
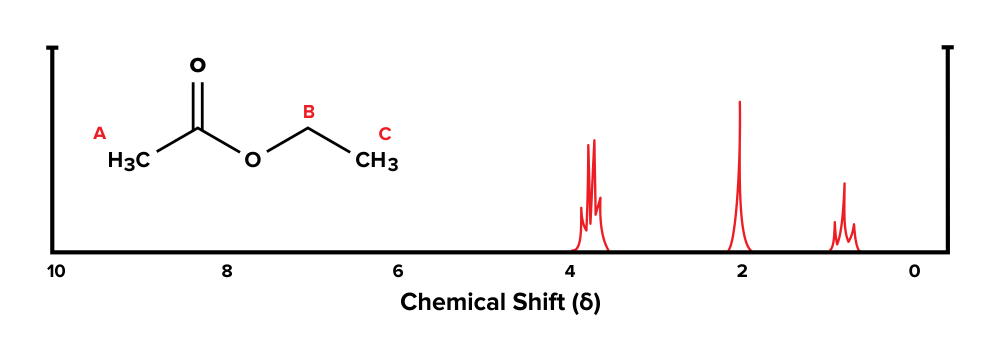
Figure: An NMR spectrum of a known molecule.
Let's get-go with functional group A. In that location are no hydrogen atoms on adjacent carbons that are within three bonds. Using the n + 1 formula, we would await 0 + 1 = i subpeaks that are grouped together. Therefore, the singlet at effectually 2 ppm corresponds with group A.
Next, let's expect at group B. At that place are three hydrogen atoms on the carbon direct adjacent to this group. Using the north + 1 formula, nosotros would expect 3 + 1 = iv subpeaks that are grouped together on the spectrum. Thus, the multiplet at around 4 ppm corresponds with this group.
Finally, group C has two adjacent hydrogen atoms in group B. Afterwards plugging in the numbers to the n + 1 formula, we would look 2 + 1 = 3 subpeaks grouped together. Therefore, the triplet at effectually 1 ppm corresponds with group C.
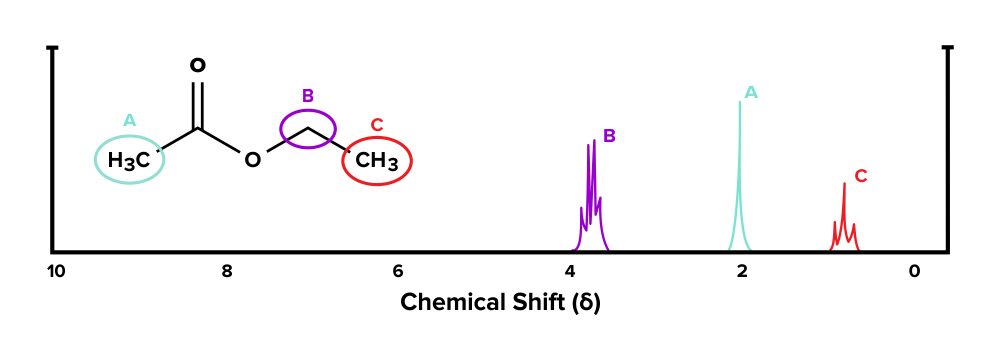
Acknowledgements: Sahil Chawla
Part 4: Loftier-yield terms
Infrared (IR) spectroscopy: allows one to identify fundamental functional groups using infrared light; detects specific frequencies at which known covalent bonds vibrate
Percentage transmittance: amount of infrared calorie-free that passes through a molecular sample; listed on the y-axis of an IR spectrum
Wavenumber: analog for frequency seen in IR spectra; measured in cm-1 and shown on the x-axis of an IR spectrum
Fingerprint region: region within 1500 to 500 cm-1 of an IR spectrum that contains peaks unique to a certain compound
Nuclear magnetic resonance (NMR) spectroscopy: provides identification of the structure of a molecule using an practical magnetic field that impacts the alignment of diminutive nuclei
Tetramethylsilane (TMS): standard used in NMR spectroscopy
Downfield: towards the left on an NMR spectrum; greater chemical shift (δ)
Upfield: towards the correct on an NMR spectrum; bottom chemic shift (δ)
Deshielding: occurs when electron-withdrawing groups pull electrons away from the core of an atom, making the atom more susceptible to an practical magnetic field
Shielding: occurs when electron-donating groups increment the electron density virtually the core of an cantlet, reducing its susceptibility to an applied magnetic field
Splitting: occurs when hydrogens fastened to adjacent carbon atoms interfere with i some other's magnetic environment
Doublet: in NMR spectra, a peak with 2 subpeaks
Triplet: in NMR spectra, a peak with three subpeaks
Multiplet: in NMR spectra, a peak with iv or more subpeaks
Part 5: Passage-based questions and answers
The World Health Organization's List of Essential Medicines is a directory of medications that have been deemed by the scientific community to be vital for any health arrangement. Among these medications are household medications such as acetaminophen. Acetaminophen is ordinarily used to care for fevers and pain.
Several students are interested in determining the structure of acetaminophen using spectroscopy. Spectroscopy is a laboratory technique that uses electromagnetic radiation and its interactions with thing to determine cardinal characteristics of compounds. This includes functional groups and the types of bonds nowadays in a molecule. Ii common spectroscopy techniques include infrared spectroscopy and nuclear magnetic resonance spectroscopy.
First, the students took a sample of acetaminophen and irradiated information technology with infrared light. A spectrometer was used to detect the frequencies at which absorption occurred. The resulting data is shown in Effigy ane.
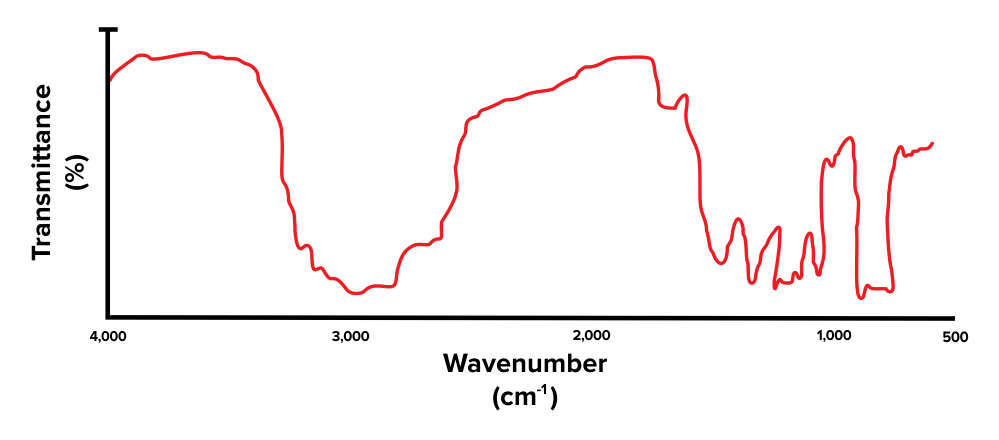
Figure 1: Resulting absorbance spectrum for acetaminophen
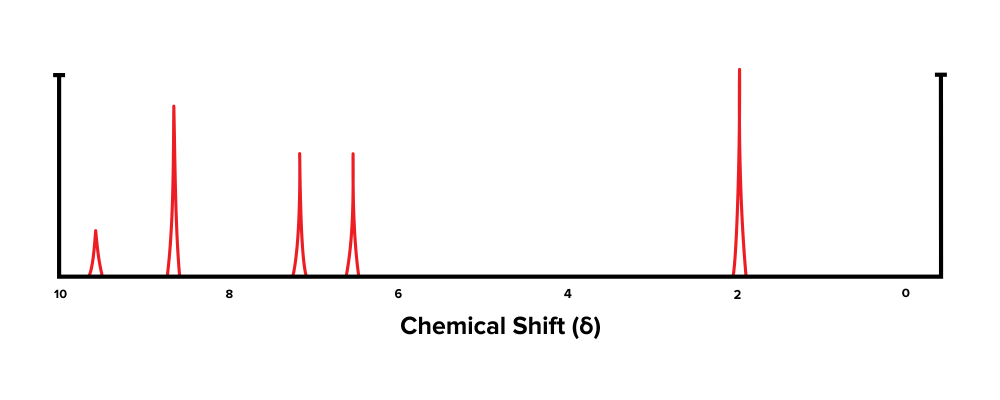
Next, the students took a sample of the drug and subjected information technology to NMR spectroscopy. The data from this process is shown in Figure 2.
Question 1: Which of the following is the range for the fingerprint region of an IR spectrum?
A) 4000 – 400 cm-ane
B) 2000 – 0 cm-1
C) 1500 – 500 cm-one
D) 4000 – 500 cm-ane
Question two: The wide pinnacle at around ~3,000 cm-1 found in Effigy 1 indicates the presence of which functional group?
A) Carbonyl
B) Carboxylic acid
C) Alcohol
D) Amino
Question 3: Which of the following statements most accurately characterizes NMR spectroscopy?
A) It uses a magnetic field to measure the mass of each individual atom in a chemical compound
B) Information technology uses a magnetic field to manipulate the alignment of atomic nuclei
C) It allows researchers to make up one's mind key functional groups but not the overall construction of a molecule
D) Information technology separates molecules based on their accuse
Question iv: Which of the following functional groups is virtually likely to generate the furthest downfield chemic shift in Figure 2?
A) TMS
B) sp3 hybridized carbon
C) Carboxylic acid
D) Aldehyde
Question 5: Based on the information in the passage, which of the following is most probable not constitute in acetaminophen?
A) Effluvious carbons
B) sp2 hybridized carbon
C) Alcohol
D) Carboxylic acid
Answer key for passage-based questions
- Respond choice C is right. The fingerprint region is the special range that is unique to a certain compound. Information technology spans the region of wavenumbers from 1500 to 500 cm-1 (pick C is correct).
- Answer pick C is right. A broad top at around ~3,000 cm-1 corresponds with the presence of an -OH bond, and thus an alcohol grouping (choice C is right). A precipitous peak at 1,750 cm-1 corresponds with the presence of a carbonyl group (choice A is wrong). Broad peaks at three,000 cm-1 and ane,750 cm-1 indicate the presence of both of these functional groups and thus propose the presence of a carboxylic acid (pick B is wrong). A abrupt peak at around ~3,300 cm-1 corresponds with the presence of an amino grouping (pick D is incorrect).
- Answer choice B is right. Nuclear magnetic resonance spectroscopy, or NMR spectroscopy, allows researchers to make up one's mind the overall structure of a molecule (choice C is incorrect). This is accomplished by applying a magnetic field that manipulates the alignment of atomic nuclei (choices A and D are incorrect).
- Answer choice D is right. The most downfield chemical shift is the one most to the left. In Figure 1, this is the peak just below 10 ppm, which has been generated past an aldehyde functional group (choice D is right). Past definition, TMS has a chemical shift of 0 ppm (choice A is incorrect). An sp3 carbon generates a chemic shift around δ=0 – 2 ppm (choice B is wrong). Carboxylic acrid carbons generate a chemic shift around δ=x - xiii ppm (option C is wrong).
- Answer choice D is correct. The resulting NMR spectrum contains peaks between 6 – 9 ppm, which indicates the presence of aromatic carbons. These carbons are sp2 hybridized (choices A and B are incorrect). The broad peak at effectually ~3,000 cm-i in the IR spectrum indicates the presence of an alcohol group (option C is incorrect). Thus, past procedure of elimination, a carboxylic acid is about probable to be absent in the structure of acetaminophen (choice D is correct).
Function 6: Standalone questions and answers
Question one: Which of the following near accurately characterizes IR spectroscopy?
A) It uses ultraviolet light to detect the presence of specific functional groups
B) It uses infrared light to observe specific frequencies at which covalent bonds vibrate
C) It uses an applied magnetic field to measure the alignment of atomic nuclei
D) It uses infrared calorie-free to measure the mass of the dissimilar atoms nowadays in a molecule
Question 2: What is tetramethylsilane?
A) A standard used to calibrate NMR spectra
B) A denaturing amanuensis necessary for NMR spectroscopy
C) A fluorescent tag that allows ane to detect the presence of a compound of involvement
D) None of the above
Question 3: Which of the following splitting patterns would exist observed in an NMR spectrum of ethanol?
A) Ane doublet and 1 triplet
B) Two doublets and one singlet
C) Two doublets and one triplet
D) One multiplet and one triplet
Question 4: What is the expected chemical shift of the functional group shown below?
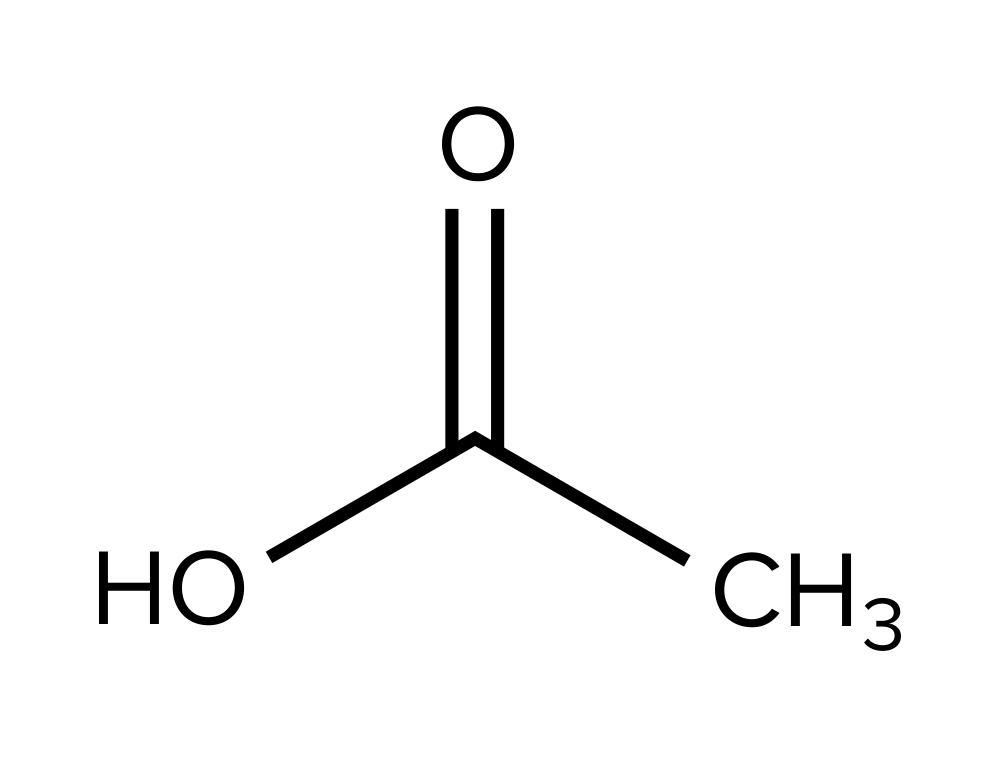
A) 0 – 2
B) 6 – 9
C) ix – 10
D) 10 – 13
Question 5: Which of the following best describes the occurrence of deshielding?
A) When electron-donating groups make the nucleus of an atom more than susceptible to the practical magnetic field
B) When electron-withdrawing groups make the nucleus of an atom more than susceptible to the practical magnetic field
C) When electron-donating groups make the nucleus of an cantlet less susceptible to the applied magnetic field
D) When electron-withdrawing groups make the nucleus of an atom less susceptible to the applied magnetic field
Answer key for standalone questions
-
Reply selection B is correct. Infrared spectroscopy uses infrared lite sources at various frequencies (choices A and C are wrong). This allows a researcher to make up one's mind the frequencies that crusade particular covalent bonds to vibrate and stretch (choice D is incorrect).
-
Answer choice A is correct. Tetramethylsilane, or TMS, is a chemic standard used to calibrate NMR spectra. It has a chemical shift of 0 ppm (choices B, C, and D are incorrect).
-
Answer selection D is correct. The methyl group (-CH₃) at the end of the ethanol molecule is adjacent to ii hydrogens. Using our north+1 formula, nosotros would await two + 1 = 3 subpeaks together. Thus, a triplet should exist present (choice B is incorrect). The hydrogens institute on the other carbon are adjacent to the three hydrogens on the methyl group. This gives us 3 + i = 4 subpeaks grouped together. Therefore, a multiplet should also be present (choices A and C are wrong).
-
Answer choice D is right. The functional group shown is a carboxylic acid group, which has a chemic shift of effectually δ=x – 13 ppm (choice D is correct). An sp³ carbon would result in a chemic shift of δ=0 – 2 ppm (choice A is wrong). An aromatic carbon would outcome in a chemical shift of δ=6 – 9 ppm (selection B is incorrect). Finally, an aldehyde grouping would issue in a chemic shift of δ=9 – ten ppm (selection C is wrong).
-
Answer option B is correct. Deshielding occurs when electron-withdrawing groups pull electrons away from an atom'southward nucleus (choices A and C are incorrect). This makes the cantlet's protons more than susceptible to the applied magnetic field (choice D is incorrect).
jeffersonpand1948.blogspot.com
Source: https://www.shemmassianconsulting.com/blog/nmr-ir-spectroscopy-mcat
0 Response to "How Much Do You Need to Know About Mass Spectroscopy for Mcat"
Postar um comentário